Table of Contents
Summary: Explore what clinical investigations nowadays have to say about stem cell therapy for spinal cord injury (SCI). Starting from safety trials to functional recovery outcomes, new evidence spotlights various potential advantages and significant limitations of regenerative medicine approaches.
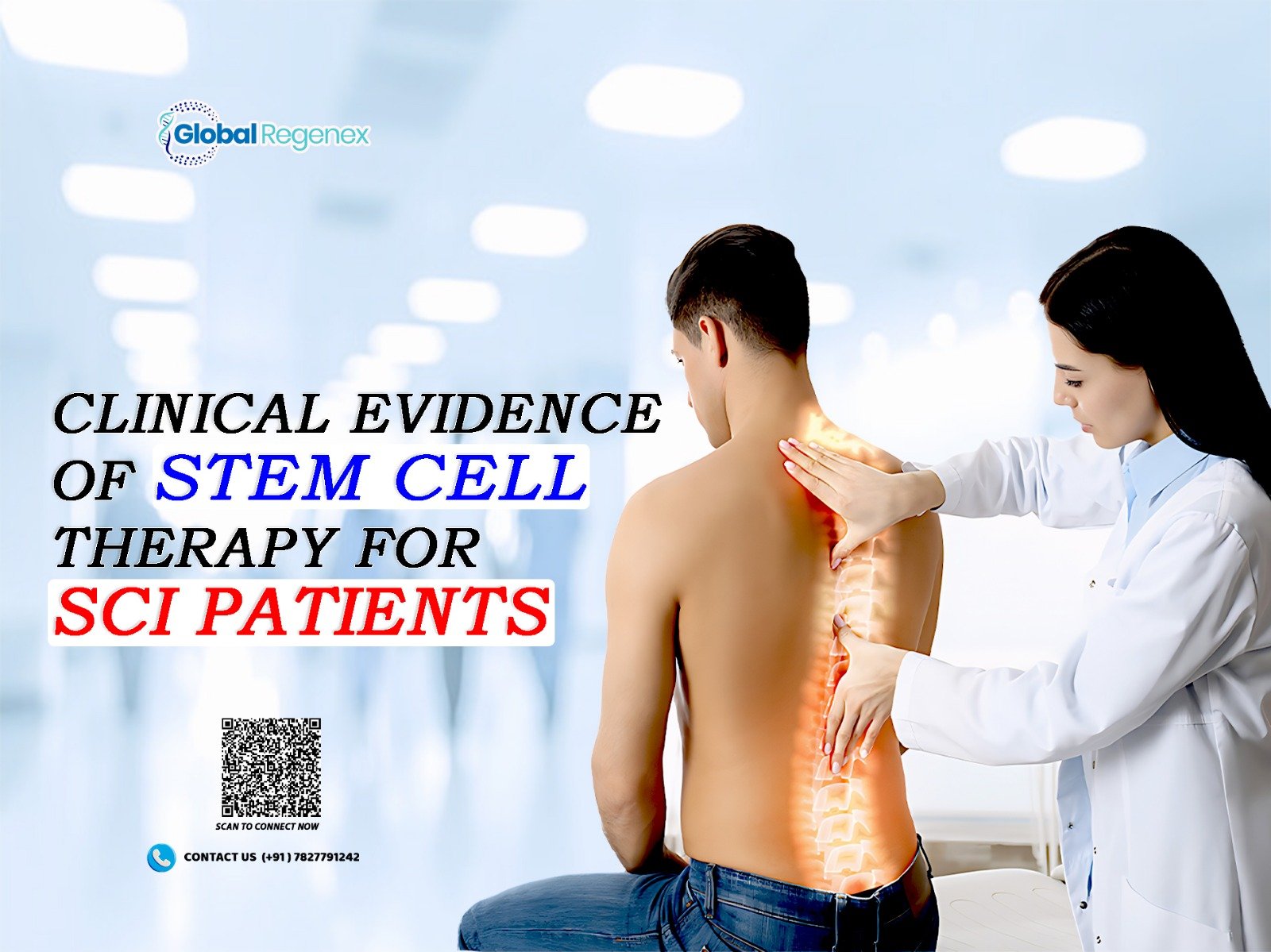
Spinal cord injury (SCI) leads to loss of muscle control and sensing either partially or completely below the area that has been damaged. To put it simply, the brain and body can no longer send signals back and forth because of the injury whether it’s an accident, trauma or a disease. The usual way to treat it is by surgery, physical therapy and other related therapies, which help to stop the injury from worsening and also make the best out of the existing functions. However, the problem still remains that we don’t know how to replace the lost nerve cells in the tissue.
That is why regenerative medicine is brought into the picture. Scientists are conducting trials to see if stem cells can be used to regenerate nerves, help with the inflammation, and lead to better almost normal functions in some of the patients with spinal cord injuries. But what does clinical evidence actually show? Keep reading.
Understanding Spinal Cord Injury
Spinal cord injury (SCI) is commonly a result of traumatic events, such as road accidents, falls, sports injuries, and acts of violence. However, tumors, infections, and degenerative diseases can also cause it.
The harm resulting from SCI is typically two fold:
- Primary Injury: The first traumatic event that causes mechanical damage to spinal cord tissue.
- Secondary Injury: The gradual inflammation, oxidative stress, and cell death that cause a further decline of the neurological condition.
The nerve cells in the central nervous system have a very limited capacity to regenerate, therefore, the recovery process is complicated.
Why Are Stem Cells Being Studied?
As conventional treatments and surgeries can give patients temporary relief. However, it does not help in long-term healing. Fortunately, stem cells are considered as supportive treatment for various conditions due to regenerative abilities and neuroprotective properties.
They may contribute to:
- Inflammation reduction
- Survival of nerve cells
- Promoting remyelination
- Improvement of the spinal cord microenvironment
- Stimulation of repair mechanisms
Instead of just replacing dying neurons, stem cells may help create conditions conducive to recovery.
How Stem Cells Are Being Studied for Spinal Cord Injury
Scientists researching stem cell therapy for SCI have been focusing on designing to really tap into multiple mechanisms of regeneration rather than focusing on a single repair route.
Neuroprotection
- MSCs secrete a variety of neurotrophic factors that can protect neurons from further degeneration.
- Clinical studies on humans have been quite scarce. Nevertheless, the initial studies show potential stabilization effects.
Immune Modulation
- After injury, the inflammatory response not only helps but also causes further damage.
- Stem cells may be able to control the immune system activity, thus, decreasing the production of inflammatory cytokines and scar formation.
Cellular Replacement
- Some exploratory studies involve using stem cells to derive neural precursor cells, which can then be used to repair damaged tissues of the spinal cord.
- However, the functional integration of these cells still needs to be thoroughly investigated.
Types of Stem Cells Studied in Spinal Cord Injury
Among the numerous types of stem cells that are being investigated:
Mesenchymal Stem Cells (MSCs): These are the ones that have been the most extensively studied in clinical trials mainly for their safety and immunomodulatory properties.
Neural Stem Cells: Mainly research is focused on the possibility of direct neural integration.
Umbilical Cord Derived Stem Cells: These cells are studied for their regenerative and anti-inflammatory properties.
Induced Pluripotent Stem Cells (iPSCs): At the moment primarily preclinical for the spinal cord regeneration research.
Every one of them has different advantages and brings different issues in research.
What Clinical Trials Have Shown So Far
Clinical research on the use of stem cells for SCI therapy has mainly been concentrated on the safety and initial functional outcomes.
The major findings from the trials reported include:
- Good Safety Profiles: A number of early, phase trials suggest that stem cell transplantation appears to be generally safe among patients in controlled medical environments.
- Neurological Recovery in Some Patients: Some research has detected increases in sensory or motor scores, especially in cases of incomplete spinal cord injury.
- Injury Subacute Better Outcomes: There is a little bit of evidence that patients treated within a few months of their injury may respond better than patients with long, term injuries.
- Functional Gains of a Small Scale: Improvements are usually partial rather than being significant. Achievements can be in terms of increased muscle strength, sensation, or bladder control.
It should be remembered that the outcomes differ dramatically from study to study depending on the severity of the injury, the timing, and the type of cells used.
The Bottom Line
Stem cell therapy is one of the most actively explored areas of regenerative medicine for spinal cord injury. Clinical trials have shown encouraging safety profiles and potential functional benefits in carefully selected patients, especially by neuroprotection and immunomodulation. Nevertheless, full recovery of the nervous system is still a scientific issue. As research is constantly developing, organized consultation with the help of Global Regenex assists patients in exploring new possibilities responsibly.
Frequently Asked Questions
Q1. Is stem cell therapy a cure for spinal cord injury?
Ans. No, as the research has progressed stem cell therapy is still in its earlier stages.
Q2. What kind of improvements have been shown in clinical trials?
Ans. There have been partial improvements in motor or sensory function in carefully selected patients.
Q3. Are stem cell therapies for SCI safe?
Ans. Early clinical trials suggest that it is safe, but long-term results are being developed.
Q4. What kind of stem cells are most often used for SCI?
Ans. Mesenchymal stem cells are most often used because of their immunomodulatory effects.
Q5. Why is consultation important before pursuing therapy?
Ans. Consulting with regenerative medicine expert before undertaking the stem cell therapy is important to know your eligibility and suitable treatment.
Reference Links
National Institute of Neurological Disorders and Stroke (NINDS) – Spinal Cord Injury Overview
https://www.ninds.nih.gov/health-information/disorders/spinal-cord-injury
PubMed Central – Stem Cell Therapy in Spinal Cord Injury
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6775579/
Frontiers in Cellular Neuroscience – MSCs in Spinal Cord Injury
https://www.frontiersin.org/articles/10.3389/fncel.2020.00021/full
International Society for Stem Cell Research (ISSCR) – Clinical Translation Guidelines
https://www.isscr.org/policy/guidelines-for-stem-cell-research-and-clinical-translation